TB Prevention
A. Prevention1
Negative Pressure Rooms:
CDC recommends that suspect TB patients be placed under Airborne Infection Isolation (AII) and placed in negative pressure rooms. Here, the air pressure inside the patient's
room is lower than the air pressure in the adjacent outside corridors (anteroom, hallway,
etc.). Consequently, outside air is "sucked" into room when a door is opened and inside
room air does not enter the adjacent spaces. Patient room air is usually exhausted
or recirculated in the same room after being "scrubbed" by filtering through a high efficiency particulate air (HEPA) filtration system. 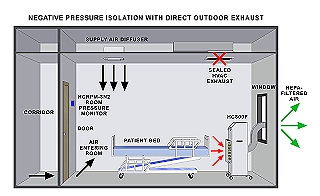
Negative pressure room (http://abatement.com/images/healthcare/hc800f_neg_pressure.gif)
"The use of personal respiratory protection is also indicated for persons entering these rooms"
Guidelines for Environmental Infection Control in Health-Care Facilities
Respirators:
Use of approved particulate filter respirator protection is an essential element of TB transmission prevention. These masks are typically disposable respirators that are commonly referred to as “N95s"
National Institute for Occupational Safety and Health (NIOSH) certifies three categories of non-powered air purifying N95 respirators based on filtering efficiency. All three categories are acceptable for use against TB:
- Type 100 (99.97% efficient)
- Type 99 (99% efficient)
- Type 95 (95% efficient)
Additional qualifications include:
- Mask's ability to filter 1 micron in size in the unloaded state with a filter efficiency greater than 95%.
- Masks must be fit tested in a reliable way to obtain face-seal leakage of less than 10%.
Healthcare workers must wear NIOSH certified respirators when:
- Entering rooms housing individuals with suspected or confirmed TB disease;
- Performing high hazard procedures on persons who have suspected or confirmed TB disease
- Emergency response employees or others must transport in a closed vehicle, an individual with suspected or confirmed TB disease.
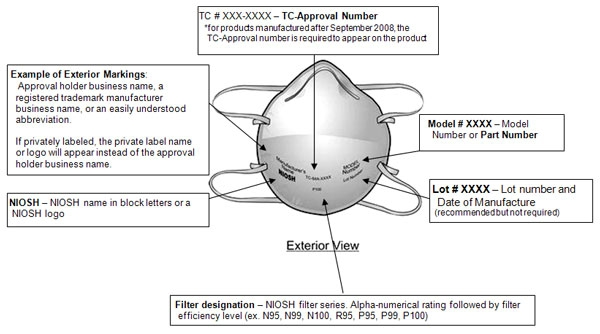
Sample of a generic filtering facepiece respirator with appropriate markings
1Information was extensively taken from Overview of Enforcement for Occupational Exposure to Tuberculosis.
More information on NIOSH-approved masks.
B. TB Testing
Method:
A Mantoux tuberculosis skin test (TST) is the primary screening method for TB infections. A TST is performed by an intradermal injection of a small amount (0.1 ml) of tuberculin purified protein derivative (PPD) into the inner forearm surface. A "good" injection results in a pale elevation of the skin (6-10 mm dia.) called a wheal. Reactions are read between 48-72 hours after inoculation. You will receive another TST if you do not do not return within 72 hours.
The area of induration (raised, hardened area or swelling) is measured in millimeters. Erythema (redness, flair) is not included in this measurement. Two factors determine the interpretation of test results:
- Induration measurement in millimeters
- Person’s risk of being infected with TB & of progression to disease if infected
A TST of 15mm or greater is considered positive in individuals without any risk factors. A TST of less than 15mm is considered positive for individuals
with known risk factors. For example an HIV-infected person with a TST of at least
5mm is considered positive. Similarly, an intravenous (IV) drug user would be
considered positive at a TST of at least 10mm.
A history of prior vaccination with bacille Calmette-Guerin (BCG) does not preclude TST.
All positive TST results require follow-up by a licensed healthcare provider before an individual can enter an at-risk healthcare setting. The Alabama Department of Public Health specifies the type of follow-up testing for a student with a positive TST. Instructions for follow-up testing (e.g., chest X-ray) are listed in this document, which is also attachment A of the CAHP - TB Surveillance & Training Program.
Different FDA-approved serological methods are methods are available to meet this requirement, if a routine chest X-ray is not indicated (e.g., possible pregnancy). Two TB whole-blood test (WBT) called Interferon-Gamma Release Assays (IGRAs) are currently available to diagnose TB infection.
- QuantiFERON®-TB Gold In-Tube test (QFT-GIT)
- T-SPOT®.TB test (T-Spot)
Requirements:
All CAHP Category I personnel required to obtain a baseline TST* before entering an at-risk facility. Annual (12-month) follow-up testing is required for all Category I personnel.
All CAHP students are required to obtain a baseline tuberculosis skin test (TST) before entering their professional program component. Student training in excess of 12-months while in an at-risk setting require additional TST. Screening frequency may be increased as specified by the individual clinical site. Category II students later reclassified as Category I must receive TST before entering an at-risk clinical site.
A two-step TB skin test is normally accomplished before entering low and/or medium risk settings. This test involves two separate TST. Here, a primary TST (1st step) is performed. Upon a negative reading it is is followed 1-3 weeks later by a second TST (2nd step). This is the recommended practice since individuals with latent (dormant) TB infections can demonstrate a negative one-step TST result when tested years after the initial infection.
Sources/Costs:
TST is available from USA Student Health Services, county public health departments, or your personal healthcare provider. TB WBT may also be offered by these same agencies. Generally, the cost for WBT is greater than that of a chest X-ray.
Initial/annual TST is provided at no cost for only Category I faculty/staff. All costs associated with TST or required follow-up testing for a positive TST is solely borne by the student.
Vaccination:
TB (BCG) vaccination is not commonly performed in the US. For more information go to the CDC website.
C. Exposure Reporting
The following protocol shall be used in all cases of a potential exposure:
- Follow local facility employee health/infection control office TB post-exposure policy or contact county public heath office if neither of the former entities is readily available.
- Inform CAHP department clinical preceptor/clinical site coordinator of the exposure.
- Perform recommended post-exposure testing (chest X-ray or TB whole blood test).
- Follow-up testing is performed at 8 weeks post-exposure.
- Document actions taken on an Evaluation of Circumstances Surrounding an Exposure Incident Form (ECP - Appendix G).
The original report form shall be retained in the faculty/staff or student's permanent record.
You will now need to complete a quiz provided by your department biosafety training
coordinator.